MEet the elements
Complete a magnet card about this video.
Radioactive Decay
1. What is radioactive decay?
2. Define one becquerel (Bq).
3. What are the different types of radioactive decay?
4. Contrast the types of radioactive decay.
5. Do all atoms decay at the same rate? Give a few examples to support your answers.
6. The decay chain of of Uranium-238 results in what final atom?
7. Is all radiation dangerous? Explain.
8. Why is radioactive decay important to geologists?
9. Why is radioactive decay important to physicians?
10. YouTube search for Pumpcast News, Part 1 - The Tonight Show with Jay Leno
1. What is radioactive decay?
2. Define one becquerel (Bq).
3. What are the different types of radioactive decay?
4. Contrast the types of radioactive decay.
5. Do all atoms decay at the same rate? Give a few examples to support your answers.
6. The decay chain of of Uranium-238 results in what final atom?
7. Is all radiation dangerous? Explain.
8. Why is radioactive decay important to geologists?
9. Why is radioactive decay important to physicians?
10. YouTube search for Pumpcast News, Part 1 - The Tonight Show with Jay Leno
Atomic Theory
Go to the following video found HERE
1. Which of the scientist mentioned in this video are also included in the above "History of the Atom Timeline?" (ignore/do not include the last four scientists included in the "Modern Quantum Cloud Model post 1930".)
2. What does the video say Democritus thought matter was made of?
3. Who made the "plum pudding" model of an atom?
4. Who proved the "plum pudding" model wrong?
5. What were the 3 parts to Rutherford's experiment?
Go to the following video found HERE
6. What is wierd/unique about visiting Rutherford's original desk drawers?
7. What happened to 1 in every 8,000 particles shot at the gold foil?
8. Because most particles passed through the foil, Rutherford was able to assume that most of atoms is made up of what?
9. Because 1 in every 8,000 bounced back, Rutherford was able to assume what about atoms?
10. According to Rutherford, if the nucleus of an atom where represented by an apple...how far away would the electrons be?
Use google to answer #11
11. What award did Henri Bacquerel win? In what year did he win it, and who did he share it with?
12. What did Bacquerel discover by accident?
13. What is radioactivity?
Use the EPA's website http://www.epa.gov/rpdweb00/understand/
14. What are the type main categories of radiation? Give an example of each.
15. Which of these two categories is more dangerous to life and why?
1. Which of the scientist mentioned in this video are also included in the above "History of the Atom Timeline?" (ignore/do not include the last four scientists included in the "Modern Quantum Cloud Model post 1930".)
2. What does the video say Democritus thought matter was made of?
3. Who made the "plum pudding" model of an atom?
4. Who proved the "plum pudding" model wrong?
5. What were the 3 parts to Rutherford's experiment?
Go to the following video found HERE
6. What is wierd/unique about visiting Rutherford's original desk drawers?
7. What happened to 1 in every 8,000 particles shot at the gold foil?
8. Because most particles passed through the foil, Rutherford was able to assume that most of atoms is made up of what?
9. Because 1 in every 8,000 bounced back, Rutherford was able to assume what about atoms?
10. According to Rutherford, if the nucleus of an atom where represented by an apple...how far away would the electrons be?
Use google to answer #11
11. What award did Henri Bacquerel win? In what year did he win it, and who did he share it with?
12. What did Bacquerel discover by accident?
13. What is radioactivity?
Use the EPA's website http://www.epa.gov/rpdweb00/understand/
14. What are the type main categories of radiation? Give an example of each.
15. Which of these two categories is more dangerous to life and why?
ALKENES
Visit the website below, and write or print out your answers and turn in with your name on them.http://chemwiki.ucdavis.edu/Organic_Chemistry/Hydrocarbons/Alkenes/Naming_the_Alkenes
1. What is the difference between an alkene and a cycloalkene?
2. How many carbons are in Eicosene?
3. Under "The Basic Rules" What should you do after you name "Give the lowest possible number to the Carbon Carbon double bond?"
Go to this link https://www.khanacademy.org/science/organic-chemistry/alkenes-alkynes/naming-alkenes/v/naming-alkenes-examples
4. What is the name of the first compound Khan names in this video?
5. Give the condensed formula for this compound he first names.
6. Around 2 min and 55 sec Khan names a second alkene...What is special or unique about this one?
7. What is the name of the second alkene Khan names (from #6 above)
8. How does Khan's way of naming (more specifically numbering) of alkenes, differ from ours in class?
9. Around 6 minutes, Khan begins to name another compound, What is strange about this one?
10. what is the name of this compound.
11. Draw the structural formula for 2,5-dimethyl-3-trans hexene12. Give the condensed formula for 2,5-dimethyl-3-trans hexene
Visit the website below, and write or print out your answers and turn in with your name on them.http://chemwiki.ucdavis.edu/Organic_Chemistry/Hydrocarbons/Alkenes/Naming_the_Alkenes
1. What is the difference between an alkene and a cycloalkene?
2. How many carbons are in Eicosene?
3. Under "The Basic Rules" What should you do after you name "Give the lowest possible number to the Carbon Carbon double bond?"
Go to this link https://www.khanacademy.org/science/organic-chemistry/alkenes-alkynes/naming-alkenes/v/naming-alkenes-examples
4. What is the name of the first compound Khan names in this video?
5. Give the condensed formula for this compound he first names.
6. Around 2 min and 55 sec Khan names a second alkene...What is special or unique about this one?
7. What is the name of the second alkene Khan names (from #6 above)
8. How does Khan's way of naming (more specifically numbering) of alkenes, differ from ours in class?
9. Around 6 minutes, Khan begins to name another compound, What is strange about this one?
10. what is the name of this compound.
11. Draw the structural formula for 2,5-dimethyl-3-trans hexene12. Give the condensed formula for 2,5-dimethyl-3-trans hexene

Google search “Gold bacteria Nature” and click on the link below:
http://www.nature.com/news/gold-digging-bacterium-makes-precious-particles-1.12352
1. When was the first evidence that bacteria can thrive off gold, discovered?
2. What is the name of the gold-thriving bacteria?
3. What is the second bacteria that can utilize gold called?
4. What is the difference between how/where the two different species of bacteria accumulate and utilize gold?
5. Describe what delftibactin is.
6. What are the two ways a “microbe assisted gold rush” might occur?
7. If 1 ml of the bacteria is able to produce 4.2 X 1024 atoms of gold, how many grams of gold could the 15 ml of bacteria produce.
Google search “ted lessons mole” and click on the link below:
http://ed.ted.com/lessons/daniel-dulek-how-big-is-a-mole-not-the-animal-the-other-one
or youtube search and the link is www.youtube.com/watch?v=TEl4jeETVmg
8. What is the name of the scientist credited with the idea of 6.02 x 10 23 molecules?
9. How many ml are in a mole of water?
10. How many donuts are in a mole of donuts?
11. Besides a dozen, list three more conversions the video mentions around 3:45.
Google search “TED lessons alien life” and click on the link below:
http://www.youtube.com/watch?v=I2apGYUX7Q0&safety_mode=true&persist_safety_mode=1&safe=active
12. Why can’t we see evidence of alien life?
13. Find two interesting or intriguing youtube videos and describe them in 2-3 sentences each.
http://www.nature.com/news/gold-digging-bacterium-makes-precious-particles-1.12352
1. When was the first evidence that bacteria can thrive off gold, discovered?
2. What is the name of the gold-thriving bacteria?
3. What is the second bacteria that can utilize gold called?
4. What is the difference between how/where the two different species of bacteria accumulate and utilize gold?
5. Describe what delftibactin is.
6. What are the two ways a “microbe assisted gold rush” might occur?
7. If 1 ml of the bacteria is able to produce 4.2 X 1024 atoms of gold, how many grams of gold could the 15 ml of bacteria produce.
Google search “ted lessons mole” and click on the link below:
http://ed.ted.com/lessons/daniel-dulek-how-big-is-a-mole-not-the-animal-the-other-one
or youtube search and the link is www.youtube.com/watch?v=TEl4jeETVmg
8. What is the name of the scientist credited with the idea of 6.02 x 10 23 molecules?
9. How many ml are in a mole of water?
10. How many donuts are in a mole of donuts?
11. Besides a dozen, list three more conversions the video mentions around 3:45.
Google search “TED lessons alien life” and click on the link below:
http://www.youtube.com/watch?v=I2apGYUX7Q0&safety_mode=true&persist_safety_mode=1&safe=active
12. Why can’t we see evidence of alien life?
13. Find two interesting or intriguing youtube videos and describe them in 2-3 sentences each.
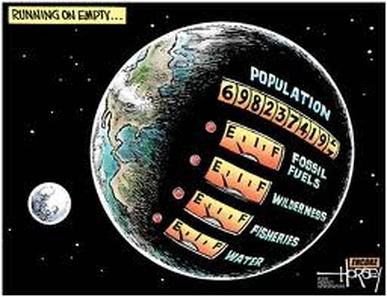
Petroleum and hydrocarbons
1. Look at the above picture. What do you think the artist's point of view about oil and drilling? Do you agree or disagree with some or all of this?
Visit the below link and answer the first 5 questions
http://www.petroleum.co.uk/chemistry/
2. What is the most prolific hydrocarbon found in petroleum?
3. Is all petroleum found everywhere in the world the same? Why or why not?
4. Describe the difference between the composition of gasoline and jet fuel.
5. Use google to look up the chemical formulas for your answer to #3. Which fuel (gas or jet fuel) contain more carbon. Why do you think this is so?
6. Click on the tab on the left titled "other uses of petroleum"...pick two usages that you did not expect to see and describe how petroleum is used in them.
Watch the following video
http://www.ted.com/talks/lisa_margonelli_the_political_chemistry_of_oil.html
7. Describe what the speaker means by calling oil "a stew of hydrocarbon molecules" (at 1 min 30 sec)
8. What about oil's chemistry makes it so disastrous?
9. At around 3 minutes, what does she compare the viscosity of oil to?
10. At around 5 minutes, what does she claim gas pumps are designed to look like? Why is this?
Visit the below link and answer the first 5 questions
http://www.petroleum.co.uk/chemistry/
2. What is the most prolific hydrocarbon found in petroleum?
3. Is all petroleum found everywhere in the world the same? Why or why not?
4. Describe the difference between the composition of gasoline and jet fuel.
5. Use google to look up the chemical formulas for your answer to #3. Which fuel (gas or jet fuel) contain more carbon. Why do you think this is so?
6. Click on the tab on the left titled "other uses of petroleum"...pick two usages that you did not expect to see and describe how petroleum is used in them.
Watch the following video
http://www.ted.com/talks/lisa_margonelli_the_political_chemistry_of_oil.html
7. Describe what the speaker means by calling oil "a stew of hydrocarbon molecules" (at 1 min 30 sec)
8. What about oil's chemistry makes it so disastrous?
9. At around 3 minutes, what does she compare the viscosity of oil to?
10. At around 5 minutes, what does she claim gas pumps are designed to look like? Why is this?